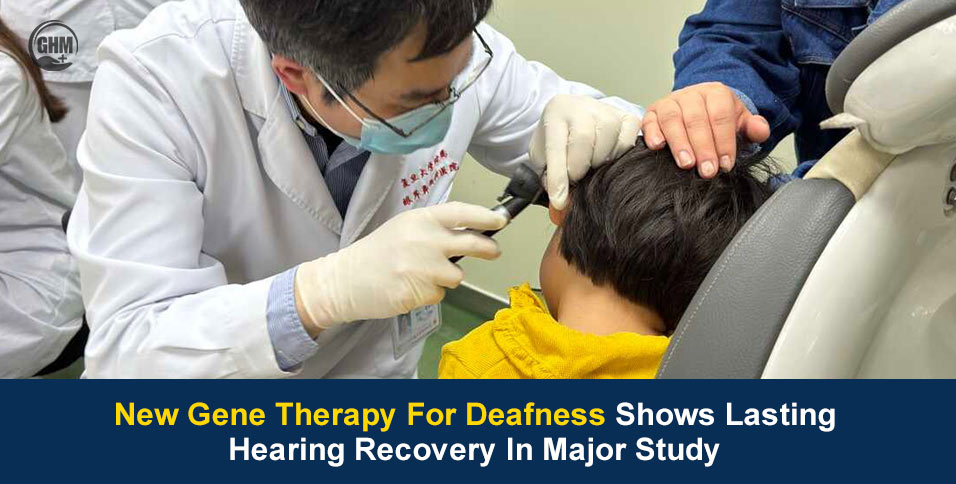
A trial on new gene therapy for deafness is offering fresh hope to families affected by inherited hearing loss after researchers reported strong and lasting results in one of the largest studies of its kind. Published in Nature, the international study found that many participants born with a rare genetic deafness, a condition called DFNB9, experienced meaningful hearing recovery after treatment.
According to the researchers, 90% of treated participants showed measurable hearing improvement during follow-up. The study tracked patients for as long as 2.5 years, suggesting the benefits may be durable rather than temporary.
This new gene therapy for deafness aims to treat the root genetic cause of hearing loss rather than only helping people manage symptoms with hearing aids or implants. While the therapy remains experimental, experts say the results could reshape the future of hearing care.
Largest Hearing Loss Gene Therapy Study So Far
The clinical trial included 42 patients between infancy and adulthood across multiple medical centers. All participants had hearing loss caused by mutations in the OTOF gene, which is linked to a rare inherited condition called DFNB9 (Autosomal Recessive Deafness 9).
According to the scientists, the hearing organ was delivered directly into the cochlea, the inner ear. The treatment used a harmless viral carrier to insert a working copy of the faulty gene.
Researchers wrote in the Nature study that hearing thresholds improved significantly after treatment, with many patients also gaining better speech recognition over time.
This makes the new gene therapy for deafness one of the most promising examples yet of hearing restoration from gene therapy.
What Is the OTOF Gene In Hearing?
The OTOF gene contains instructions for making otoferlin, a protein needed for sound signals to move from inner ear hair cells to the auditory nerve.
When the gene does not work properly, the ear may detect sound but fail to pass those signals to the brain efficiently. This can cause severe or profound deafness from birth.
Because the cause is clear and linked to one gene, researchers believed OTOF gene therapy could be a strong target for hearing treatment.
This clinical trial of gene therapy treatment for inherited deafness tries to replace the missing instructions so the ear can function more normally.
How Did New Gene Therapy Treat Inherited Deafness?
This hearing loss trial did not cure all deafness, and not yet, but there is a 90% possibility.
The new gene therapy for deafness was tested only in patients with deafness caused by specific OTOF mutations, aged 0.5 to 18 years. It does not currently apply to every type of hearing loss, such as age-related hearing decline, noise damage, infections, or many other genetic causes.
However, for the right patients, the results suggest gene therapy may partially restore hearing and speech perception. That is why many experts view it as a major milestone in the treatment of rare genetic deafness.
Some children in earlier programs began responding to voices and sounds after treatment, outcomes that were previously unlikely without devices.
Who May Benefit Most From Gene Therapy For Hearing?
The study found that younger patients often responded better than older adults to the gene therapy treatment. Researchers believe early treatment may help because the hearing system and language pathways are still developing.
Groups who may benefit most include:
- Children diagnosed early with OTOF-related deafness
- Families with confirmed genetic testing results
- Patients with congenital hearing loss therapy needs linked to single-gene mutations
- Individuals evaluated at specialist hearing centers
Experts say newborn hearing screening plus faster genetic testing could become more valuable if therapies like this move toward approval.
With this breakthrough trial, pediatric hearing loss treatment could become one of the most important future uses of gene therapy.
Safety Results and Next Steps
Researchers reported that the therapy was generally well-tolerated. No major dose-limiting safety issues were identified during the primary monitoring period.
Some participants experienced temporary side effects, which researchers said require continued observation. Long-term follow-up remains essential for any gene-based therapy.
The next step in new gene therapy for deafness will likely involve larger trials, longer monitoring, and regulatory review before broader use.
Conclusion
The new gene therapy for deafness is still in the clinical research stage and has not become a standard treatment worldwide.
More studies are needed to confirm long-term safety, effectiveness, ideal dosing, and which patients benefit most.
Still, momentum in the field is growing. Recent advances in other conditions, including experimental work in GLP-1 gene therapy and research into Huntington’s disease treatment, show how rapidly genetic medicine is expanding.
If future trials confirm success, similar strategies could be developed for other inherited hearing disorders. For families living with congenital deafness, that possibility is powerful.